高同型半胱氨酸(专业版)
高同型半胱氨酸血症(HCY,简称高血同)是指血浆中同型半胱氨酸含量增高,这是由甲硫氨酸代谢障碍引起的。
其他名称:高血同;高同型半胱氨酸血症
英文名称:High Homocysteine,Hyperhomocysteinemia
据报道,高血同是我国高血压患者特有风险因素,约75%的高血压患者伴有高血同,导致卒中风险增加10倍以上。此外,HCY还可导致妊娠并发症和习惯性流产或早产等。
同型半胱氨酸通过两种途径代谢:再甲基化和转硫化。
随着对血管损伤累积,可导致或促进多种与血管相关的疾病,包括:
调整饮食与生活方式
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于减少HCY的营养和草本补充剂,主要包括如下:
1.维生素B族:
B族维生素是用于治疗高同型半胱氨酸水平的主要治疗剂。许多研究证实了它们单独或组合降低高同型半胱氨酸水平的能力,一些试验表明,它们具有降低中风和痴呆风险的临床益处。一般来说,维生素B12和叶酸的联合使用比单独使用更有效。维生素B6和B2在降低同型半胱氨酸治疗中的益处也有报道1,2。
1).叶酸:
叶酸存在于许多植物性食物中,但经常在烹饪或加工过程中丢失或降解3。此外,摄入量通常较低,尤其是在老年人中4。叶酸缺乏与认知能力下降、抑郁和神经系统疾病有关5,6。也有证据表明叶酸缺乏与阿尔茨海默病相关的表观遗传紊乱相联系7。
需要足够的叶酸状态来将同型半胱氨酸重新甲基化为甲硫氨酸。研究发现,每天补充0.5-5mg叶酸可以使同型半胱氨酸水平降低约25%。由于叶酸缺乏与神经管出生缺陷之间的密切关系,美国于1998年开始强制强化谷物制品。从那时起,中年人的同型半胱氨酸水平下降了约7%8。
多项研究发现,补充叶酸可以改善认知功能,尤其是同型半胱氨酸水平高的人9。临床试验的荟萃分析报告称,通过叶酸治疗降低同型半胱氨酸的水平,可将中风风险平均降低10%,将所有心血管事件风险平均降低4%。这些益处与同型半胱氨酸的降低程度相关,在基线叶酸水平较低的人群中更为明显8,10。一项对49项随机对照试验的荟萃分析发现,在降低血压、降低心血管事件和中风风险方面,服用叶酸加降压药物比单独服用降压药物更有效在患有高血压的受试者中。服用叶酸超过12周的人和同型半胱氨酸水平下降超过25%的人的益处最大11。叶酸对代谢性疾病患者的帮助可能较小,因为对2型糖尿病和其他代谢性疾病参与者的试验的荟萃分析发现,叶酸改善了胰岛素敏感性,但对血压、空腹血糖水平、血糖控制或脂质水平没有影响12,13。
关于:选择活性叶酸而不是叶酸的重要性
叶酸补充剂一般是用合成叶酸(蝶酰谷氨酸)制成的,叶酸在大多数人体内可转化为可使用的活性形式,即5-甲基四氢叶酸(5-MTHF,或称活性叶酸)。然而,相当大比例的人群有一种或多种遗传变异,干扰叶酸向5-MTHF的有效转化14。
L-甲基叶酸是一些叶酸补充剂中常用的一种形式(5-MTHF,活性叶酸)。这意味着L-甲基叶酸形式不需要MTHFR酶来产生用于同型半胱氨酸代谢的功能性叶酸15。带有这些低效率MTHFR基因的人从L-甲基叶酸中获益比从合成叶酸补充剂中获益更多16。即使是MTHFR基因状态正常的人,与传统的合成叶酸相比,对L-甲基叶酸形式的反应也更好17-21。
重要的是,已经证明高剂量的合成叶酸可以阻止正常人和5-MTHFR基因变异人群的叶酸代谢22,23。幸运的是,补充L-甲基叶酸完全避免了这个问题。
2). 维生素B12:
维生素B12(简称钴胺素),通常以各种形式存在于食物和补充剂中。摄入后,钴胺素被转运到细胞中以转化为甲基钴胺,这是同型半胱氨酸代谢所需的活性形式24。维生素 B12以氰钴胺、羟钴胺或甲钴胺的形式存在,由于在消化道中的吸收率低,通常作为肌内注射给药;然而,有一些证据表明,每天口服1000–2000mcg的维生素B12,可以有效地使血液中的维生素B12水平正常化25,26。
检测B12状态最常见的测试是血清总B12;然而,由于血液中只有约6-20%的B12具有代谢活性,即使B12总水平在正常范围内的人也可能存在活性不足的情况27-29。虽然正常范围一般为160–950pg/ml,但需要至少540pg/ml的B12水平来防止同型半胱氨酸水平上升30。B12水平低于正常范围的一半,加上同型半胱氨酸水平升高,表明代谢性B12缺乏27。
维生素B12和叶酸之间的密切关系使得很难区分它们的独立缺乏和治疗效果。维生素B12缺乏会导致功能性叶酸缺乏,因为它会将叶酸“困住”为5-甲基四叶酸,从而阻止其发挥其他功能。另一方面,补充叶酸可以通过使红细胞的变化正常化来“掩盖”B12缺乏,而红细胞的改变通常是B12缺乏的早期迹象31。
临床证据表明,每天补充高达1000mcg的B12可以安全地降低B12缺乏症患者的同型半胱氨酸水平,并且随着剂量的增加,这种效果会增强32,33。血清B12水平低与高血压和神经系统疾病有关,而维持较高的B12水平似乎还能防止脑组织萎缩,并可能有助于预防抑郁症,特别是在老年人中34,35。在降低同型半胱氨酸的治疗中加入B12可以提高治疗效果并降低脑卒中风险1,27。12,25值得注意的是,一些证据表明,长期高剂量的氰钴胺可能对肾病患者有害;因此,甲钴胺和羟钴胺是维生素B12治疗的优选形式27。
3). 维生素B6:
维生素B6(吡哆醇)是细胞中140多种反应的辅助因子,包括同型半胱氨酸的再甲基化和转硫。尽管维生素B6在食品中广泛存在,但在西方国家,多达31%的非养老院老年人的维生素B6摄入量较低4。低B6状态会导致同型半胱氨酸积累,并降低甲基化反应的SAMe可用性,导致神经递质合成减少以及相关的情绪和神经问题36。很少有研究独立于叶酸和B12来研究维生素B6在同型半胱氨酸相关疾病中的作用,但一项荟萃分析确定,摄入更多的维生素B6与患冠心病的风险较低相关37。
维生素B6仅以磷酸化形式(5-磷酸吡哆醛)具有活性,在某些情况下,补充这种形式可能对改善B6状态更有效38。大多数补充剂含有吡哆醇,它很容易通过细胞膜转运并被磷酸化24。长期服用过高剂量的维生素B6会导致类似B6缺乏的神经症状,但一些证据表明,只有吡哆醇形式与这种毒性作用有关39。
4). 维生素B2:
维生素B2(核黄素)的摄入量和血液水平在老年人中通常较低4,40。B2是许多细胞酶的辅因子,包括两种参与同型半胱氨酸再甲基化的酶:亚甲基四氢叶酸还原酶(MTHFR,有助于激活叶酸作为甲基供体)和甲硫氨酸合成酶还原酶(MTRR,与维生素B12一起促进同型半胱氨酸的再甲基化)41。
同型半胱氨酸水平随着核黄素摄入量和血液水平的下降而升高,补充B2对同型半胱氨酸代谢有积极影响,特别是在具有影响叶酸循环的MTHFR基因变体的个体中42,43。此外,无论MTHFR的基因型如何,单独使用叶酸治疗高同型半胱氨酸水平都会消耗维生素B2,降低叶酸治疗的潜在疗效44。
5).维生素B组合:
维生素B2、B6、B12和叶酸通过其在同型半胱氨酸代谢和甲基化途径中的相互依赖作用而密不可分。它们之间密切的功能关系可以从它们的缺乏症共有的许多症状这一事实中得到说明。不管它们之间的相互关系如何,大多数研究都独立研究了B族维生素降低同型半胱氨酸的能力,其中最重视叶酸,其次分别是B12和B6,很少关注B2。补充全量的B族维生素的潜在益处在很大程度上尚未被探索33。
在一项随机对照试验中,在两年内每天补充500mcg B12、800mcg叶酸和20mg B6,可将轻度认知障碍和高基线同型半胱氨酸水平患者的灰质萎缩(最易患阿尔茨海默病的脑组织)减少7倍45。
一项对照试验证明了联合维生素B补充剂优于单独叶酸。在该试验中,104名患有高血压和高血同的参与者每天接受5mg叶酸或每天补充400mcg叶酸(以活性叶酸计)、5mcg B12、3mg B6和2.4mg B2,以及12.5mg锌和250mg甜菜碱。叶酸组的平均同型半胱氨酸水平从22.6µmol/L降至14.3µmol/L,联合用药组从21.5µmol/L降至10.0µmol/L。此外,超过55%的服用联合补充剂的人的同型半胱氨酸水平<10µmol/L,研究作者认为这是理想的46。
2. 胆碱和甜菜碱:
胆碱是一种存在于许多食物中的营养成分,如蛋黄、乳制品、肉类、花生、十字花科蔬菜、坚果和种子、全谷物和大豆。它是细胞膜的重要结构成分,既是神经递质乙酰胆碱的前体,也是脑组织不可分割的一部分。胆碱也可用于制造甜菜碱或三甲基甘氨酸,起到甲基供体的作用47。甜菜碱是将同型半胱氨酸重新甲基化为甲硫氨酸的辅助因子,尤其是在缺乏足够叶酸的情况下47。甜菜碱除了在体内产生外,还存在于海鲜、小麦胚芽和麸皮、甜菜和菠菜等食物中48。
胆碱和甜菜碱摄入不足会导致SAMe的产生减少和同型半胱氨酸的积累增加。这种失衡损害了细胞中的甲基化过程,导致细胞功能崩溃和DNA的表观遗传变化49。
甜菜碱在调节同型半胱氨酸水平方面发挥着关键作用,尤其是在低叶酸、低B12和高蛋氨酸条件下50,51。尽管它存在于许多和各种食物中,但NHANES(美国国家健康和营养检测调查)发现,只有不到11%的美国人达到了胆碱的推荐摄入量,即男性每天550mg,女性每天425mg52。
每天补充1000–6000mg的甜菜碱已被证明可以降低血液同型半胱氨酸水平,并减缓摄入甲硫氨酸后同型半胱氨酸的增加53-55。在一项针对23名运动员的安慰剂对照试验中,那些在为期六周的运动训练计划中每天服用2500mg甜菜碱的运动员,其尿同型半胱氨酸硫内酯的增加较低,它一种同型半胱氨酸,已知对血管有毒性作用,并干扰正常蛋白质的产生和功能56,57。然而,需要注意的是,大量摄入胆碱和甜菜碱对心血管疾病风险和死亡率的影响尚不清楚58,59。
3.欧米伽3脂肪酸:
Omega-3脂肪酸似乎与B族维生素协同作用,促进健康的同型半胱氨酸代谢,降低与高同型半胱氨酸水平相关的疾病风险,包括心血管和神经系统疾病60。多项随机对照试验表明,鱼油(EPA和DHA)可以降低同型半胱氨酸水平,添加维生素B12、B6和叶酸可以增强其作用61。
ω-3脂肪酸对心血管和神经健康的有益作用可能取决于是否有足够的同型半胱氨酸代谢。例如,一项针对阿尔茨海默病患者的临床试验分析发现,DHA和EPA治疗仅能改善同型半胱氨酸水平低于11.7µmol/L患者的认知功能62。此外,高水平的ω-3脂肪酸可以防止同型半胱氨酸毒性,并增强B族维生素的益处。
在一项研究中,高同型半胱氨酸水平与低ω-3状态的老年受试者大脑β-淀粉样蛋白(阿尔茨海默病风险的标志物)增加有关,但与高omega-3水平的老年人无关63。其他临床试验发现,B族维生素减缓轻度认知障碍患者脑组织萎缩和改善认知功能的能力取决于血液中ω-3脂肪酸的充足水平,尤其是DHA(二十二碳六烯酸)64,65。
4. S-腺苷蛋氨酸:
S-腺苷蛋氨酸(SAMe,或称S腺苷甲硫氨酸)是许多细胞过程中的重要甲基供体,包括表观遗传基因修饰和神经递质合成。由于高同型半胱氨酸水平通常是同型半胱氨酸转化为甲硫氨酸的不良结果,甲硫氨酸是SAMe的前体,SAMe通常随着同型半胱氨酸的积累而耗尽66。这可能是高同型型半胱氨酸水平与抑郁等心理情绪状况之间关系的一个促成因素,以及在与叶酸代谢受损相关的MTHFR基因变体携带者中发现的更高的抑郁症风险67,68。
临床证据表明,SAMe可能有助于治疗抑郁症,包括同型半胱氨酸水平升高的人68,69。一份病例报告描述了SAMe对一名被发现患有MTHFR基因突变的焦虑症患者的益处:甲基化B12和叶酸治疗对缓解症状无效,直到添加SAMe,每天两次,400mg70。尽管人们担心补充SAMe可能会增加同型半胱氨酸的产生,但一项针对重度抑郁症患者的试验发现,每天800–1600mg的SAMe持续六周并不能提高同型半胱氨酸水平71。
5. N-乙酰半胱氨酸:
NAC是半胱氨酸的一种来源,可在体内用于制造体内重要的抗氧化剂—谷胱甘肽。通过增加谷胱甘肽的产生和降低氧化应激,人们认为NAC可能有助于减轻过量同型半胱氨酸的一些毒性作用。此外,NAC似乎降低了同型半胱氨酸水平72,73。
在一项为期八周的随机对照试验中,60名患有高同型半胱氨酸水平和冠状动脉疾病的参与者,与安慰剂相比,每天600mg NAC与每天5mg叶酸对降低同型半胱氨酸水平的有效性相当74。在中年男性中进行的安慰剂对照试验发现,连续四周每天补充1800mg NAC可平均降低近12%的同型半胱氨酸。NAC还能降低血压,尤其是那些胆固醇和甘油三酯水平高的患者75。在另一项试验中,30名同型半胱氨酸水平高、阿尔茨海默病或相关疾病的患者接受了一种补充剂的治疗,该补充剂提供了未公开剂量的叶酸(如5-MTHF)、维生素B12(甲钴胺)和NAC,持续时间从2.5个月到34.6个月不等。与未接受补充剂治疗的类似患者相比,接受B维生素/NAC组合治疗的患者的脑组织萎缩显著减少76。
6.牛磺酸:
牛磺酸是一种非必需氨基酸,与蛋氨酸、半胱氨酸和同型半胱氨酸一样,也是硫的来源。牛磺酸可以由体内的半胱氨酸制成,对血管、神经、代谢和肌肉骨骼健康产生有益影响77,78。临床前证据表明,补充牛磺酸可以降低高同型半胱氨酸水平,保护心脏和血管细胞免受同型半胱氨酸诱导的损伤79-81。在一项初步试验中,22名中年妇女每天服用3g牛磺酸,持续四周。结果导致平均同型半胱氨酸水平从8.5µmol/L降至7.6µmol/L82。即使在血液同型半胱氨酸浓度>125µmol/L的受试者中,由于一种名为同型胱氨酸尿症的遗传疾病,补充牛磺酸也能改善血管功能83。
7. 镁:
镁可能有助于对抗高同型半胱氨酸水平的负面影响。众所周知,镁可以预防心血管疾病,如高血压、动脉粥样硬化、心律失常、冠状动脉疾病和心力衰竭。大量研究表明,镁的摄入与降低心脏病发作和中风风险之间存在关联84,85。研究进一步表明,镁可能有助于保护认知功能和神经健康86。
一项实验室研究的结果表明,低镁状态可能会加剧同型半胱氨酸引发的细胞内镁损失87。另一项对实验室培养的血管细胞的研究发现,同型半胱氨酸增加了引发与斑块形成相关的结构变化的化合物产生;然而,在细胞环境中添加镁可以减轻同型半胱氨酸的这种致动脉粥样硬化作用88。在实验室小鼠中,镁也被证明可以逆转同型半胱氨酸对心律的不利影响89。
更多可点击其个性化的综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
目前尚没有专门治疗高同型半胱氨酸血症的药物
参考文献:
1. Hankey GJ. B vitamins for stroke prevention. Stroke Vasc Neurol. 2018;3(2):51-58.
2. Miller JW. Proton Pump Inhibitors, H2-Receptor Antagonists, Metformin, and Vitamin B-12 Deficiency: Clinical Implications. Adv Nutr. 2018;9(4):511s-518s.
3. Delchier N et al. Mechanisms of folate losses during processing: diffusion vs. heat degradation. Food Chem. 2014;157:439-447.
4. Ter Borg S et al. Micronutrient intakes and potential inadequacies of community-dwelling older adults: a systematic review. The British journal of nutrition. 2015;113(8):1195-1206.
5. Sobczynska-Malefora A et al. Laboratory assessment of folate (vitamin B9) status. Journal of clinical pathology. 2018;71(11):949-956.
6. Baroni L et al. Association Between Cognitive Impairment and Vitamin B12, Folate, and Homocysteine Status in Elderly Adults: A Retrospective Study. J Alzheimers Dis. 2019;70(2):443-453.
7. Robinson N et al. Alzheimer's disease pathogenesis: Is there a role for folate? Mech Ageing Dev. 2018;174:86-94.
8. Li Y et al. Folic Acid Supplementation and the Risk of Cardiovascular Diseases: A Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. 2016;5(8).
9. Enderami A et al. The effects and potential mechanisms of folic acid on cognitive function: a comprehensive review. Neurol Sci. 2018;39(10):1667-1675.
10. Tian T et al. Folic Acid Supplementation for Stroke Prevention in Patients With Cardiovascular Disease. The American journal of the medical sciences. 2017;354(4):379-387.
11. Wang WW et al. A Meta-Analysis of Folic Acid in Combination with Anti-Hypertension Drugs in Patients with Hypertension and Hyperhomocysteinemia. Frontiers in pharmacology. 2017;8:585.
12. Akbari M et al. The Effects of Folate Supplementation on Diabetes Biomarkers Among Patients with Metabolic Diseases: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Hormone and metabolic research. 2018;50(2):93-105.
13. Tabrizi R et al. The effects of folate supplementation on lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Diabetes Metab Syndr. 2018;12(3):423-430.
14. GARD. MTHFR gene variant. NIH. rarediseases.info.nih.gov Web site. https://rarediseases.info.nih.gov/diseases/10953/mthfr-gene-mutation. Published 2019.
15. Scaglione F et al. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica. 2014;44(5):480-488.
16. Servy E et al. The Methylene Tetrahydrofolate Reductase (MTHFR) isoform challenge. High doses of folic acid are not a suitable option compared to 5 Methyltetrahydrofolate treatment. Clinical Obstetrics, Gynecology and Reproductive Medicine. 2017;3:1-5.
17. Prinz-Langenohl R et al. [6S]-5-methyltetrahydrofolate increases plasma folate more effectively than folic acid in women with the homozygous or wild-type 677C-->T polymorphism of methylenetetrahydrofolate reductase. Br J Pharmacol. 2009;158(8):2014-2021.
18. Willems FF et al. Pharmacokinetic study on the utilisation of 5-methyltetrahydrofolate and folic acid in patients with coronary artery disease. Br J Pharmacol. 2004;141(5):825-830.
19. Henderson AM et al. l-5-Methyltetrahydrofolate Supplementation Increases Blood Folate Concentrations to a Greater Extent than Folic Acid Supplementation in Malaysian Women. J Nutr. 2018;148(6):885-890.
20. Venn BJ et al. Comparison of the effect of low-dose supplementation with L-5-methyltetrahydrofolate or folic acid on plasma homocysteine: a randomized placebo-controlled study. Am J Clin Nutr. 2003;77(3):658-662.
21. Lamers Y et al. Red blood cell folate concentrations increase more after supplementation with [6S]-5-methyltetrahydrofolate than with folic acid in women of childbearing age. Am J Clin Nutr. 2006;84(1):156-161.
22. Cornet D et al. High doses of folic acid induce a pseudo-methylenetetrahydrofolate syndrome. SAGE Open Med Case Rep. 2019;7:2050313x19850435.
23. Tafuri L et al. The hazards of excessive folic acid intake in MTHFR gene mutation carriers: An obstetric and gynecological perspective. Clinical Obstetrics, Gynecology and Reproductive Medicine. 2018;4:1-2.
24. Calderon-Ospina CA et al. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS neuroscience & therapeutics. 2020;26(1):5-13.
25. Wolffenbuttel BHR et al. The Many Faces of Cobalamin (Vitamin B12) Deficiency. Mayo Clin Proc Innov Qual Outcomes. 2019;3(2):200-214.
26. Wang H et al. Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency. The Cochrane database of systematic reviews. 2018;3:Cd004655.
27. Spence JD. Cardioembolic stroke: everything has changed. Stroke Vasc Neurol. 2018;3(2):76-83.
28. Roman GC et al. Epigenetic Factors in Late-Onset Alzheimer's Disease: MTHFR and CTH Gene Polymorphisms, Metabolic Transsulfuration and Methylation Pathways, and B Vitamins. International journal of molecular sciences. 2019;20(2).
29. Obeid R et al. Vitamin B12 Intake From Animal Foods, Biomarkers, and Health Aspects. Frontiers in nutrition. 2019;6:93.
30. NIH. Vitamin B12 Level. US National Library of Medicine: Medline Plus. Available at https://medlineplus.gov/ency/article/003705.htm. Last update 1/19/2018.
31. Field MS et al. Safety of folic acid. Ann N Y Acad Sci. 2018;1414(1):59-71.
32. Eussen SJ et al. Oral cyanocobalamin supplementation in older people with vitamin B12 deficiency: a dose-finding trial. Arch Intern Med. 2005;165(10):1167-1172.
33. Kennedy DO. B Vitamins and the Brain: Mechanisms, Dose and Efficacy--A Review. Nutrients. 2016;8(2):68.
34. Watson J et al. Consequences of Inadequate Intakes of Vitamin A, Vitamin B12, Vitamin D, Calcium, Iron, and Folate in Older Persons. Curr Geriatr Rep. 2018;7(2):103-113.
35. Pavlov CS et al. Neurological disorders in vitamin B12 deficiency. Terapevticheskii arkhiv. 2019;91(4):122-129.
36. Parra M et al. Vitamin B(6) and Its Role in Cell Metabolism and Physiology. Cells. 2018;7(7).
37. Jayedi A et al. Intake of vitamin B6, folate, and vitamin B12 and risk of coronary heart disease: a systematic review and dose-response meta-analysis of prospective cohort studies. Crit Rev Food Sci Nutr. 2019;59(16):2697-2707.
38. Wilson MP et al. Disorders affecting vitamin B6 metabolism. Journal of inherited metabolic disease. 2019;42(4):629-646.
39. Vrolijk MF et al. The vitamin B6 paradox: Supplementation with high concentrations of pyridoxine leads to decreased vitamin B6 function. Toxicol In Vitro. 2017;44:206-212.
40. Porter K et al. Causes, Consequences and Public Health Implications of Low B-Vitamin Status in Ageing. Nutrients. 2016;8(11).
41. Esse R, Barroso M, Tavares de Almeida I, Castro R. The Contribution of Homocysteine Metabolism Disruption to Endothelial Dysfunction: State-of-the-Art. International journal of molecular sciences. 2019;20(4).
42. Troesch B et al. Potential Links between Impaired One-Carbon Metabolism Due to Polymorphisms, Inadequate B-Vitamin Status, and the Development of Alzheimer's Disease. Nutrients. 2016;8(12).
43. Garcia-Minguillan CJ et al. Riboflavin status modifies the effects of methylenetetrahydrofolate reductase (MTHFR) and methionine synthase reductase (MTRR) polymorphisms on homocysteine. Genes Nutr. 2014;9(6):435.
44. Marashly ET et al. Riboflavin Has Neuroprotective Potential: Focus on Parkinson's Disease and Migraine. Frontiers in neurology. 2017;8:333.
45. Douaud G et al. Preventing Alzheimer's disease-related gray matter atrophy by B-vitamin treatment. Proc Natl Acad Sci U S A. 2013;110(23):9523-9528.
46. Mazza A et al. Nutraceutical approaches to homocysteine lowering in hypertensive subjects at low cardiovascular risk: a multicenter, randomized clinical trial. Journal of biological regulators and homeostatic agents. 2016;30(3):921-927.
47. Ueland PM. Choline and betaine in health and disease. Journal of inherited metabolic disease. 2011;34(1):3-15.
48. Craig SA. Betaine in human nutrition. The American Journal of Clinical Nutrition. 2004;80(3):539-549.
49. Zeisel S. Choline, Other Methyl-Donors and Epigenetics. Nutrients. 2017;9(5).
50. Obeid R. The metabolic burden of methyl donor deficiency with focus on the betaine homocysteine methyltransferase pathway. Nutrients. 2013;5(9):3481-3495.
51. Lee JE et al. Are dietary choline and betaine intakes determinants of total homocysteine concentration? Am J Clin Nutr. 2010;91(5):1303-1310.
52. Wallace TC et al. Assessment of Total Choline Intakes in the United States. J Am Coll Nutr. 2016;35(2):108-112.
53. Atkinson W et al. Dietary and supplementary betaine: effects on betaine and homocysteine concentrations in males. Nutr Metab Cardiovasc Dis. 2009;19(11):767-773.
54. Olthof MR et al. Effects of betaine intake on plasma homocysteine concentrations and consequences for health. Curr Drug Metab. 2005;6(1):15-22.
55. Steenge GR et al. Betaine supplementation lowers plasma homocysteine in healthy men and women. J Nutr. 2003;133(5):1291-1295.
56. Jakubowski H et al. Chemical biology of homocysteine thiolactone and related metabolites. Advances in clinical chemistry. 2011;55:81-103.
57. Cholewa JM et al. Effects of betaine on body composition, performance, and homocysteine thiolactone. Journal of the International Society of Sports Nutrition. 2013;10(1):39.
58. Guasch-Ferre M et al. Plasma Metabolites From Choline Pathway and Risk of Cardiovascular Disease in the PREDIMED (Prevention With Mediterranean Diet) Study. J Am Heart Assoc. 2017;6(11).
59. Meyer KA et al. Dietary Choline and Betaine and Risk of CVD: A Systematic Review and Meta-Analysis of Prospective Studies. Nutrients. 2017;9(7).
60. Rizzo G et al. The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions. Biomolecules. 2020;10(2).
61. Dawson SL et al. A combination of omega-3 fatty acids, folic acid and B-group vitamins is superior at lowering homocysteine than omega-3 alone: A meta-analysis. Nutr Res. 2016;36(6):499-508.
62. Jerneren F et al. Homocysteine Status Modifies the Treatment Effect of Omega-3 Fatty Acids on Cognition in a Randomized Clinical Trial in Mild to Moderate Alzheimer's Disease: The OmegAD Study. J Alzheimers Dis. 2019;69(1):189-197.
63. Hooper C et al. Cross-Sectional Associations of Total Plasma Homocysteine with Cortical beta-Amyloid Independently and as a Function of Omega 3 Polyunsaturated Fatty Acid Status in Older Adults at Risk of Dementia. The journal of nutrition, health & aging. 2017;21(10):1075-1080.
64. Jerneren F et al. Brain atrophy in cognitively impaired elderly: the importance of long-chain omega-3 fatty acids and B vitamin status in a randomized controlled trial. Am J Clin Nutr. 2015;102(1):215-221.
65. Oulhaj A et al. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J Alzheimers Dis. 2016;50(2):547-557.
66. Fu Y et al. Hyperhomocysteinaemia and vascular injury: advances in mechanisms and drug targets. Br J Pharmacol. 2018;175(8):1173-1189.
67. Bhatia P et al. Homocysteine excess: delineating the possible mechanism of neurotoxicity and depression. Fundamental & clinical pharmacology. 2015;29(6):522-528.
68. Papakostas GI et al. Folates and S-adenosylmethionine for major depressive disorder. Can J Psychiatry. 2012;57(7):406-413.
69. Karas Kuzelicki N. S-Adenosyl Methionine in the Therapy of Depression and Other Psychiatric Disorders. Drug Dev Res. 2016;77(7):346-356.
70. Anderson S et al. Anxiety and Methylenetetrahydrofolate Reductase Mutation Treated With S-Adenosyl Methionine and Methylated B Vitamins. Integrative medicine (Encinitas, Calif). 2016;15(2):48-52.
71. Mischoulon D et al. Bioavailability of S-adenosyl methionine and impact on response in a randomized, double-blind, placebo-controlled trial in major depressive disorder. The Journal of clinical psychiatry. 2012;73(6):843-848.
72. Aydin AF et al. N-Acetylcysteine supplementation decreased brain lipid and protein oxidations produced by experimental homocysteine thiolactone exposure: Relevance to neurodegeneration. Pathophysiology. 2018;25(2):125-129.
73. Kondakci G et al. The effect of N-acetylcysteine supplementation on serum homocysteine levels and hepatic and renal oxidative stress in homocysteine thiolactone-treated rats. Arch Physiol Biochem. 2017;123(2):128-133.
74. Yilmaz H et al. Effects of folic acid and N-acetylcysteine on plasma homocysteine levels and endothelial function in patients with coronary artery disease. Acta cardiologica. 2007;62(6):579-585.
75. Hildebrandt W et al. Oral N-acetylcysteine reduces plasma homocysteine concentrations regardless of lipid or smoking status. Am J Clin Nutr. 2015;102(5):1014-1024.
76. Shankle WR et al. CerefolinNAC Therapy of Hyperhomocysteinemia Delays Cortical and White Matter Atrophy in Alzheimer's Disease and Cerebrovascular Disease. J Alzheimers Dis. 2016;54(3):1073-1084.
77. Schaffer S et al. Effects and Mechanisms of Taurine as a Therapeutic Agent. Biomolecules & therapeutics. 2018;26(3):225-241.
78. Jakaria M et al. Taurine and its analogs in neurological disorders: Focus on therapeutic potential and molecular mechanisms. Redox biology. 2019;24:101223.
79. Zulli A et al. High dietary taurine reduces apoptosis and atherosclerosis in the left main coronary artery: association with reduced CCAAT/enhancer binding protein homologous protein and total plasma homocysteine but not lipidemia. Hypertension. 2009;53(6):1017-1022.
80. Nonaka H et al. Taurine prevents the decrease in expression and secretion of extracellular superoxide dismutase induced by homocysteine: amelioration of homocysteine-induced endoplasmic reticulum stress by taurine. Circulation. 2001;104(10):1165-1170.
81. Zhang Z et al. Taurine ameliorated homocysteine-induced H9C2 cardiomyocyte apoptosis by modulating endoplasmic reticulum stress. Apoptosis. 2017;22(5):647-661.
82. Ahn CS. Effect of taurine supplementation on plasma homocysteine levels of the middle-aged Korean women. Adv Exp Med Biol. 2009;643:415-422.
83. Van Hove JLK et al. Biomarkers of oxidative stress, inflammation, and vascular dysfunction in inherited cystathionine beta-synthase deficient homocystinuria and the impact of taurine treatment in a phase 1/2 human clinical trial. Journal of inherited metabolic disease. 2019;42(3):424-437.
84. Zhao L et al. Quantitative Association Between Serum/Dietary Magnesium and Cardiovascular Disease/Coronary Heart Disease Risk: A Dose-Response Meta-analysis of Prospective Cohort Studies. Journal of cardiovascular pharmacology. 2019;74(6):516-527.
85. Rosique-Esteban N et al. Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients. 2018;10(2).
86. Kirkland AE et al. The Role of Magnesium in Neurological Disorders. Nutrients. 2018;10(6).
87. Li W et al. Extracellular magnesium regulates effects of vitamin B6, B12 and folate on homocysteinemia-induced depletion of intracellular free magnesium ions in canine cerebral vascular smooth muscle cells: possible relationship to [Ca2+]i, atherogenesis and stroke. Neurosci Lett. 1999;274(2):83-86.
88. Guo H et al. Effects of folic acid and magnesium on the production of homocysteine-induced extracellular matrix metalloproteinase-2 in cultured rat vascular smooth muscle cells. Circulation journal. 2006;70(1):141-146.
89. Soni CV et al. Hyperhomocysteinemia Alters Sinoatrial and Atrioventricular Nodal Function: Role of Magnesium in Attenuating These Effects. Cell Biochem Biophys. 2016;74(1):59-65.
美国国立公众健康网
www.medlineplus.gov
美国国立心脏、肺、血液研究所
http://www.nhlbi.nih.gov
美国家庭医生学会
http://familydoctor.org
加拿大心血管协会
http://www.ccs.ca
加拿大家庭医生学会
http://www.cfpc.ca
免责声明和安全信息
英文名称:High Homocysteine,Hyperhomocysteinemia
定义
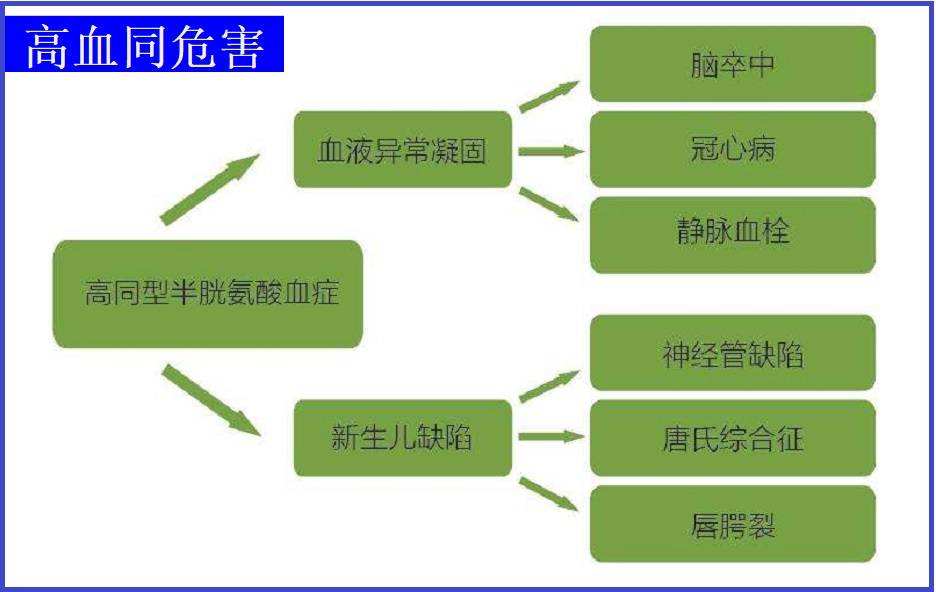
高同型半胱氨酸血症(HCY,简称高血同)是指血浆中同型半胱氨酸含量增高,这是由甲硫氨酸代谢障碍引起的。HCY可刺激动脉血管内壁而引起损伤,导致炎症和斑块形成,使动脉血流受阻。研究表明,高血同是心脑血管疾病的一个独立和重要的危险因素,可导致冠心病、老年痴呆、中风和脑萎缩等。据报道,高血同是我国高血压患者特有风险因素,约75%的高血压患者伴有高血同,导致卒中风险增加10倍以上。此外,HCY还可导致妊娠并发症和习惯性流产或早产等。
病因
同型半胱氨酸是一种含硫氨基酸,为甲硫氨酸和半胱氨酸代谢过程中产生的中间产物。正常情况下,同型半胱氨酸在体内能被分解代谢而维持在较低水平。然而,由于遗传和环境原因引起代谢障碍,导致HCY。同型半胱氨酸通过两种途径代谢:再甲基化和转硫化。
- 再甲基化需要叶酸和B12辅酶。活性叶酸(L甲基叶酸)与维生素B12(甲钴胺)作为甲基供体,将同型半胱氨酸转化为甲硫氨酸(或蛋氨酸)。
- 转硫过程需要吡哆醛-5'-磷酸参与,B6辅酶。
风险因素
许多因素可导致高同型半胱氨酸水平:- 叶酸、维生素B6、维生素B12等缺乏或不足
- 高蛋氨酸饮食,包括红肉和乳制品
- 年龄增长,营养吸收不良或摄入不足
- 某些药物,包括消胆胺、考来替泊、左旋多巴、二甲双胍、甲氨蝶呤和烟酸等
- 吸烟
- 过量咖啡
- 过量饮酒
- 肥胖
- 遗传变异,代谢叶酸能力受损
症状
作为心血管疾病的独立危险因素,HCY早期不会引起体征或症状。随着对血管损伤累积,可导致或促进多种与血管相关的疾病,包括:
- 血管疾病,如冠心病、外周动脉疾病和颈部动脉疾病等
- 脑血管疾病,如中风、痴呆症
- 糖尿病合并症,如糖尿病合并高血压、糖尿病肾病和糖尿病视网膜病变等
- 血栓病,如动脉血栓、深静脉血栓等。
- 妊娠并发症,包括如:
- 流产、早产
- 先兆子痫
- 新生儿缺陷
并发症
未经控制的HCY可能导致如下并发症:疗法
综合选项可包括如下:调整饮食与生活方式
- 健康饮食,丰富蔬菜、水果,及全谷类食物
- 限制富含蛋氨酸食物,如猪牛羊肉、乳制品
- 坚持运动锻炼
- 适度饮酒,或不饮酒
- 不抽烟,或戒烟
- 保持健康体重
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于减少HCY的营养和草本补充剂,主要包括如下:
1.维生素B族:
B族维生素是用于治疗高同型半胱氨酸水平的主要治疗剂。许多研究证实了它们单独或组合降低高同型半胱氨酸水平的能力,一些试验表明,它们具有降低中风和痴呆风险的临床益处。一般来说,维生素B12和叶酸的联合使用比单独使用更有效。维生素B6和B2在降低同型半胱氨酸治疗中的益处也有报道1,2。
1).叶酸:
叶酸存在于许多植物性食物中,但经常在烹饪或加工过程中丢失或降解3。此外,摄入量通常较低,尤其是在老年人中4。叶酸缺乏与认知能力下降、抑郁和神经系统疾病有关5,6。也有证据表明叶酸缺乏与阿尔茨海默病相关的表观遗传紊乱相联系7。
需要足够的叶酸状态来将同型半胱氨酸重新甲基化为甲硫氨酸。研究发现,每天补充0.5-5mg叶酸可以使同型半胱氨酸水平降低约25%。由于叶酸缺乏与神经管出生缺陷之间的密切关系,美国于1998年开始强制强化谷物制品。从那时起,中年人的同型半胱氨酸水平下降了约7%8。
多项研究发现,补充叶酸可以改善认知功能,尤其是同型半胱氨酸水平高的人9。临床试验的荟萃分析报告称,通过叶酸治疗降低同型半胱氨酸的水平,可将中风风险平均降低10%,将所有心血管事件风险平均降低4%。这些益处与同型半胱氨酸的降低程度相关,在基线叶酸水平较低的人群中更为明显8,10。一项对49项随机对照试验的荟萃分析发现,在降低血压、降低心血管事件和中风风险方面,服用叶酸加降压药物比单独服用降压药物更有效在患有高血压的受试者中。服用叶酸超过12周的人和同型半胱氨酸水平下降超过25%的人的益处最大11。叶酸对代谢性疾病患者的帮助可能较小,因为对2型糖尿病和其他代谢性疾病参与者的试验的荟萃分析发现,叶酸改善了胰岛素敏感性,但对血压、空腹血糖水平、血糖控制或脂质水平没有影响12,13。
关于:选择活性叶酸而不是叶酸的重要性
叶酸补充剂一般是用合成叶酸(蝶酰谷氨酸)制成的,叶酸在大多数人体内可转化为可使用的活性形式,即5-甲基四氢叶酸(5-MTHF,或称活性叶酸)。然而,相当大比例的人群有一种或多种遗传变异,干扰叶酸向5-MTHF的有效转化14。
L-甲基叶酸是一些叶酸补充剂中常用的一种形式(5-MTHF,活性叶酸)。这意味着L-甲基叶酸形式不需要MTHFR酶来产生用于同型半胱氨酸代谢的功能性叶酸15。带有这些低效率MTHFR基因的人从L-甲基叶酸中获益比从合成叶酸补充剂中获益更多16。即使是MTHFR基因状态正常的人,与传统的合成叶酸相比,对L-甲基叶酸形式的反应也更好17-21。
重要的是,已经证明高剂量的合成叶酸可以阻止正常人和5-MTHFR基因变异人群的叶酸代谢22,23。幸运的是,补充L-甲基叶酸完全避免了这个问题。
2). 维生素B12:
维生素B12(简称钴胺素),通常以各种形式存在于食物和补充剂中。摄入后,钴胺素被转运到细胞中以转化为甲基钴胺,这是同型半胱氨酸代谢所需的活性形式24。维生素 B12以氰钴胺、羟钴胺或甲钴胺的形式存在,由于在消化道中的吸收率低,通常作为肌内注射给药;然而,有一些证据表明,每天口服1000–2000mcg的维生素B12,可以有效地使血液中的维生素B12水平正常化25,26。
检测B12状态最常见的测试是血清总B12;然而,由于血液中只有约6-20%的B12具有代谢活性,即使B12总水平在正常范围内的人也可能存在活性不足的情况27-29。虽然正常范围一般为160–950pg/ml,但需要至少540pg/ml的B12水平来防止同型半胱氨酸水平上升30。B12水平低于正常范围的一半,加上同型半胱氨酸水平升高,表明代谢性B12缺乏27。
维生素B12和叶酸之间的密切关系使得很难区分它们的独立缺乏和治疗效果。维生素B12缺乏会导致功能性叶酸缺乏,因为它会将叶酸“困住”为5-甲基四叶酸,从而阻止其发挥其他功能。另一方面,补充叶酸可以通过使红细胞的变化正常化来“掩盖”B12缺乏,而红细胞的改变通常是B12缺乏的早期迹象31。
临床证据表明,每天补充高达1000mcg的B12可以安全地降低B12缺乏症患者的同型半胱氨酸水平,并且随着剂量的增加,这种效果会增强32,33。血清B12水平低与高血压和神经系统疾病有关,而维持较高的B12水平似乎还能防止脑组织萎缩,并可能有助于预防抑郁症,特别是在老年人中34,35。在降低同型半胱氨酸的治疗中加入B12可以提高治疗效果并降低脑卒中风险1,27。12,25值得注意的是,一些证据表明,长期高剂量的氰钴胺可能对肾病患者有害;因此,甲钴胺和羟钴胺是维生素B12治疗的优选形式27。
3). 维生素B6:
维生素B6(吡哆醇)是细胞中140多种反应的辅助因子,包括同型半胱氨酸的再甲基化和转硫。尽管维生素B6在食品中广泛存在,但在西方国家,多达31%的非养老院老年人的维生素B6摄入量较低4。低B6状态会导致同型半胱氨酸积累,并降低甲基化反应的SAMe可用性,导致神经递质合成减少以及相关的情绪和神经问题36。很少有研究独立于叶酸和B12来研究维生素B6在同型半胱氨酸相关疾病中的作用,但一项荟萃分析确定,摄入更多的维生素B6与患冠心病的风险较低相关37。
维生素B6仅以磷酸化形式(5-磷酸吡哆醛)具有活性,在某些情况下,补充这种形式可能对改善B6状态更有效38。大多数补充剂含有吡哆醇,它很容易通过细胞膜转运并被磷酸化24。长期服用过高剂量的维生素B6会导致类似B6缺乏的神经症状,但一些证据表明,只有吡哆醇形式与这种毒性作用有关39。
4). 维生素B2:
维生素B2(核黄素)的摄入量和血液水平在老年人中通常较低4,40。B2是许多细胞酶的辅因子,包括两种参与同型半胱氨酸再甲基化的酶:亚甲基四氢叶酸还原酶(MTHFR,有助于激活叶酸作为甲基供体)和甲硫氨酸合成酶还原酶(MTRR,与维生素B12一起促进同型半胱氨酸的再甲基化)41。
同型半胱氨酸水平随着核黄素摄入量和血液水平的下降而升高,补充B2对同型半胱氨酸代谢有积极影响,特别是在具有影响叶酸循环的MTHFR基因变体的个体中42,43。此外,无论MTHFR的基因型如何,单独使用叶酸治疗高同型半胱氨酸水平都会消耗维生素B2,降低叶酸治疗的潜在疗效44。
5).维生素B组合:
维生素B2、B6、B12和叶酸通过其在同型半胱氨酸代谢和甲基化途径中的相互依赖作用而密不可分。它们之间密切的功能关系可以从它们的缺乏症共有的许多症状这一事实中得到说明。不管它们之间的相互关系如何,大多数研究都独立研究了B族维生素降低同型半胱氨酸的能力,其中最重视叶酸,其次分别是B12和B6,很少关注B2。补充全量的B族维生素的潜在益处在很大程度上尚未被探索33。
在一项随机对照试验中,在两年内每天补充500mcg B12、800mcg叶酸和20mg B6,可将轻度认知障碍和高基线同型半胱氨酸水平患者的灰质萎缩(最易患阿尔茨海默病的脑组织)减少7倍45。
一项对照试验证明了联合维生素B补充剂优于单独叶酸。在该试验中,104名患有高血压和高血同的参与者每天接受5mg叶酸或每天补充400mcg叶酸(以活性叶酸计)、5mcg B12、3mg B6和2.4mg B2,以及12.5mg锌和250mg甜菜碱。叶酸组的平均同型半胱氨酸水平从22.6µmol/L降至14.3µmol/L,联合用药组从21.5µmol/L降至10.0µmol/L。此外,超过55%的服用联合补充剂的人的同型半胱氨酸水平<10µmol/L,研究作者认为这是理想的46。
2. 胆碱和甜菜碱:
胆碱是一种存在于许多食物中的营养成分,如蛋黄、乳制品、肉类、花生、十字花科蔬菜、坚果和种子、全谷物和大豆。它是细胞膜的重要结构成分,既是神经递质乙酰胆碱的前体,也是脑组织不可分割的一部分。胆碱也可用于制造甜菜碱或三甲基甘氨酸,起到甲基供体的作用47。甜菜碱是将同型半胱氨酸重新甲基化为甲硫氨酸的辅助因子,尤其是在缺乏足够叶酸的情况下47。甜菜碱除了在体内产生外,还存在于海鲜、小麦胚芽和麸皮、甜菜和菠菜等食物中48。
胆碱和甜菜碱摄入不足会导致SAMe的产生减少和同型半胱氨酸的积累增加。这种失衡损害了细胞中的甲基化过程,导致细胞功能崩溃和DNA的表观遗传变化49。
甜菜碱在调节同型半胱氨酸水平方面发挥着关键作用,尤其是在低叶酸、低B12和高蛋氨酸条件下50,51。尽管它存在于许多和各种食物中,但NHANES(美国国家健康和营养检测调查)发现,只有不到11%的美国人达到了胆碱的推荐摄入量,即男性每天550mg,女性每天425mg52。
每天补充1000–6000mg的甜菜碱已被证明可以降低血液同型半胱氨酸水平,并减缓摄入甲硫氨酸后同型半胱氨酸的增加53-55。在一项针对23名运动员的安慰剂对照试验中,那些在为期六周的运动训练计划中每天服用2500mg甜菜碱的运动员,其尿同型半胱氨酸硫内酯的增加较低,它一种同型半胱氨酸,已知对血管有毒性作用,并干扰正常蛋白质的产生和功能56,57。然而,需要注意的是,大量摄入胆碱和甜菜碱对心血管疾病风险和死亡率的影响尚不清楚58,59。
3.欧米伽3脂肪酸:
Omega-3脂肪酸似乎与B族维生素协同作用,促进健康的同型半胱氨酸代谢,降低与高同型半胱氨酸水平相关的疾病风险,包括心血管和神经系统疾病60。多项随机对照试验表明,鱼油(EPA和DHA)可以降低同型半胱氨酸水平,添加维生素B12、B6和叶酸可以增强其作用61。
ω-3脂肪酸对心血管和神经健康的有益作用可能取决于是否有足够的同型半胱氨酸代谢。例如,一项针对阿尔茨海默病患者的临床试验分析发现,DHA和EPA治疗仅能改善同型半胱氨酸水平低于11.7µmol/L患者的认知功能62。此外,高水平的ω-3脂肪酸可以防止同型半胱氨酸毒性,并增强B族维生素的益处。
在一项研究中,高同型半胱氨酸水平与低ω-3状态的老年受试者大脑β-淀粉样蛋白(阿尔茨海默病风险的标志物)增加有关,但与高omega-3水平的老年人无关63。其他临床试验发现,B族维生素减缓轻度认知障碍患者脑组织萎缩和改善认知功能的能力取决于血液中ω-3脂肪酸的充足水平,尤其是DHA(二十二碳六烯酸)64,65。
4. S-腺苷蛋氨酸:
S-腺苷蛋氨酸(SAMe,或称S腺苷甲硫氨酸)是许多细胞过程中的重要甲基供体,包括表观遗传基因修饰和神经递质合成。由于高同型半胱氨酸水平通常是同型半胱氨酸转化为甲硫氨酸的不良结果,甲硫氨酸是SAMe的前体,SAMe通常随着同型半胱氨酸的积累而耗尽66。这可能是高同型型半胱氨酸水平与抑郁等心理情绪状况之间关系的一个促成因素,以及在与叶酸代谢受损相关的MTHFR基因变体携带者中发现的更高的抑郁症风险67,68。
临床证据表明,SAMe可能有助于治疗抑郁症,包括同型半胱氨酸水平升高的人68,69。一份病例报告描述了SAMe对一名被发现患有MTHFR基因突变的焦虑症患者的益处:甲基化B12和叶酸治疗对缓解症状无效,直到添加SAMe,每天两次,400mg70。尽管人们担心补充SAMe可能会增加同型半胱氨酸的产生,但一项针对重度抑郁症患者的试验发现,每天800–1600mg的SAMe持续六周并不能提高同型半胱氨酸水平71。
5. N-乙酰半胱氨酸:
NAC是半胱氨酸的一种来源,可在体内用于制造体内重要的抗氧化剂—谷胱甘肽。通过增加谷胱甘肽的产生和降低氧化应激,人们认为NAC可能有助于减轻过量同型半胱氨酸的一些毒性作用。此外,NAC似乎降低了同型半胱氨酸水平72,73。
在一项为期八周的随机对照试验中,60名患有高同型半胱氨酸水平和冠状动脉疾病的参与者,与安慰剂相比,每天600mg NAC与每天5mg叶酸对降低同型半胱氨酸水平的有效性相当74。在中年男性中进行的安慰剂对照试验发现,连续四周每天补充1800mg NAC可平均降低近12%的同型半胱氨酸。NAC还能降低血压,尤其是那些胆固醇和甘油三酯水平高的患者75。在另一项试验中,30名同型半胱氨酸水平高、阿尔茨海默病或相关疾病的患者接受了一种补充剂的治疗,该补充剂提供了未公开剂量的叶酸(如5-MTHF)、维生素B12(甲钴胺)和NAC,持续时间从2.5个月到34.6个月不等。与未接受补充剂治疗的类似患者相比,接受B维生素/NAC组合治疗的患者的脑组织萎缩显著减少76。
6.牛磺酸:
牛磺酸是一种非必需氨基酸,与蛋氨酸、半胱氨酸和同型半胱氨酸一样,也是硫的来源。牛磺酸可以由体内的半胱氨酸制成,对血管、神经、代谢和肌肉骨骼健康产生有益影响77,78。临床前证据表明,补充牛磺酸可以降低高同型半胱氨酸水平,保护心脏和血管细胞免受同型半胱氨酸诱导的损伤79-81。在一项初步试验中,22名中年妇女每天服用3g牛磺酸,持续四周。结果导致平均同型半胱氨酸水平从8.5µmol/L降至7.6µmol/L82。即使在血液同型半胱氨酸浓度>125µmol/L的受试者中,由于一种名为同型胱氨酸尿症的遗传疾病,补充牛磺酸也能改善血管功能83。
7. 镁:
镁可能有助于对抗高同型半胱氨酸水平的负面影响。众所周知,镁可以预防心血管疾病,如高血压、动脉粥样硬化、心律失常、冠状动脉疾病和心力衰竭。大量研究表明,镁的摄入与降低心脏病发作和中风风险之间存在关联84,85。研究进一步表明,镁可能有助于保护认知功能和神经健康86。
一项实验室研究的结果表明,低镁状态可能会加剧同型半胱氨酸引发的细胞内镁损失87。另一项对实验室培养的血管细胞的研究发现,同型半胱氨酸增加了引发与斑块形成相关的结构变化的化合物产生;然而,在细胞环境中添加镁可以减轻同型半胱氨酸的这种致动脉粥样硬化作用88。在实验室小鼠中,镁也被证明可以逆转同型半胱氨酸对心律的不利影响89。
更多可点击其个性化的综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
目前尚没有专门治疗高同型半胱氨酸血症的药物
参考文献:
1. Hankey GJ. B vitamins for stroke prevention. Stroke Vasc Neurol. 2018;3(2):51-58.
2. Miller JW. Proton Pump Inhibitors, H2-Receptor Antagonists, Metformin, and Vitamin B-12 Deficiency: Clinical Implications. Adv Nutr. 2018;9(4):511s-518s.
3. Delchier N et al. Mechanisms of folate losses during processing: diffusion vs. heat degradation. Food Chem. 2014;157:439-447.
4. Ter Borg S et al. Micronutrient intakes and potential inadequacies of community-dwelling older adults: a systematic review. The British journal of nutrition. 2015;113(8):1195-1206.
5. Sobczynska-Malefora A et al. Laboratory assessment of folate (vitamin B9) status. Journal of clinical pathology. 2018;71(11):949-956.
6. Baroni L et al. Association Between Cognitive Impairment and Vitamin B12, Folate, and Homocysteine Status in Elderly Adults: A Retrospective Study. J Alzheimers Dis. 2019;70(2):443-453.
7. Robinson N et al. Alzheimer's disease pathogenesis: Is there a role for folate? Mech Ageing Dev. 2018;174:86-94.
8. Li Y et al. Folic Acid Supplementation and the Risk of Cardiovascular Diseases: A Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. 2016;5(8).
9. Enderami A et al. The effects and potential mechanisms of folic acid on cognitive function: a comprehensive review. Neurol Sci. 2018;39(10):1667-1675.
10. Tian T et al. Folic Acid Supplementation for Stroke Prevention in Patients With Cardiovascular Disease. The American journal of the medical sciences. 2017;354(4):379-387.
11. Wang WW et al. A Meta-Analysis of Folic Acid in Combination with Anti-Hypertension Drugs in Patients with Hypertension and Hyperhomocysteinemia. Frontiers in pharmacology. 2017;8:585.
12. Akbari M et al. The Effects of Folate Supplementation on Diabetes Biomarkers Among Patients with Metabolic Diseases: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Hormone and metabolic research. 2018;50(2):93-105.
13. Tabrizi R et al. The effects of folate supplementation on lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Diabetes Metab Syndr. 2018;12(3):423-430.
14. GARD. MTHFR gene variant. NIH. rarediseases.info.nih.gov Web site. https://rarediseases.info.nih.gov/diseases/10953/mthfr-gene-mutation. Published 2019.
15. Scaglione F et al. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica. 2014;44(5):480-488.
16. Servy E et al. The Methylene Tetrahydrofolate Reductase (MTHFR) isoform challenge. High doses of folic acid are not a suitable option compared to 5 Methyltetrahydrofolate treatment. Clinical Obstetrics, Gynecology and Reproductive Medicine. 2017;3:1-5.
17. Prinz-Langenohl R et al. [6S]-5-methyltetrahydrofolate increases plasma folate more effectively than folic acid in women with the homozygous or wild-type 677C-->T polymorphism of methylenetetrahydrofolate reductase. Br J Pharmacol. 2009;158(8):2014-2021.
18. Willems FF et al. Pharmacokinetic study on the utilisation of 5-methyltetrahydrofolate and folic acid in patients with coronary artery disease. Br J Pharmacol. 2004;141(5):825-830.
19. Henderson AM et al. l-5-Methyltetrahydrofolate Supplementation Increases Blood Folate Concentrations to a Greater Extent than Folic Acid Supplementation in Malaysian Women. J Nutr. 2018;148(6):885-890.
20. Venn BJ et al. Comparison of the effect of low-dose supplementation with L-5-methyltetrahydrofolate or folic acid on plasma homocysteine: a randomized placebo-controlled study. Am J Clin Nutr. 2003;77(3):658-662.
21. Lamers Y et al. Red blood cell folate concentrations increase more after supplementation with [6S]-5-methyltetrahydrofolate than with folic acid in women of childbearing age. Am J Clin Nutr. 2006;84(1):156-161.
22. Cornet D et al. High doses of folic acid induce a pseudo-methylenetetrahydrofolate syndrome. SAGE Open Med Case Rep. 2019;7:2050313x19850435.
23. Tafuri L et al. The hazards of excessive folic acid intake in MTHFR gene mutation carriers: An obstetric and gynecological perspective. Clinical Obstetrics, Gynecology and Reproductive Medicine. 2018;4:1-2.
24. Calderon-Ospina CA et al. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS neuroscience & therapeutics. 2020;26(1):5-13.
25. Wolffenbuttel BHR et al. The Many Faces of Cobalamin (Vitamin B12) Deficiency. Mayo Clin Proc Innov Qual Outcomes. 2019;3(2):200-214.
26. Wang H et al. Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency. The Cochrane database of systematic reviews. 2018;3:Cd004655.
27. Spence JD. Cardioembolic stroke: everything has changed. Stroke Vasc Neurol. 2018;3(2):76-83.
28. Roman GC et al. Epigenetic Factors in Late-Onset Alzheimer's Disease: MTHFR and CTH Gene Polymorphisms, Metabolic Transsulfuration and Methylation Pathways, and B Vitamins. International journal of molecular sciences. 2019;20(2).
29. Obeid R et al. Vitamin B12 Intake From Animal Foods, Biomarkers, and Health Aspects. Frontiers in nutrition. 2019;6:93.
30. NIH. Vitamin B12 Level. US National Library of Medicine: Medline Plus. Available at https://medlineplus.gov/ency/article/003705.htm. Last update 1/19/2018.
31. Field MS et al. Safety of folic acid. Ann N Y Acad Sci. 2018;1414(1):59-71.
32. Eussen SJ et al. Oral cyanocobalamin supplementation in older people with vitamin B12 deficiency: a dose-finding trial. Arch Intern Med. 2005;165(10):1167-1172.
33. Kennedy DO. B Vitamins and the Brain: Mechanisms, Dose and Efficacy--A Review. Nutrients. 2016;8(2):68.
34. Watson J et al. Consequences of Inadequate Intakes of Vitamin A, Vitamin B12, Vitamin D, Calcium, Iron, and Folate in Older Persons. Curr Geriatr Rep. 2018;7(2):103-113.
35. Pavlov CS et al. Neurological disorders in vitamin B12 deficiency. Terapevticheskii arkhiv. 2019;91(4):122-129.
36. Parra M et al. Vitamin B(6) and Its Role in Cell Metabolism and Physiology. Cells. 2018;7(7).
37. Jayedi A et al. Intake of vitamin B6, folate, and vitamin B12 and risk of coronary heart disease: a systematic review and dose-response meta-analysis of prospective cohort studies. Crit Rev Food Sci Nutr. 2019;59(16):2697-2707.
38. Wilson MP et al. Disorders affecting vitamin B6 metabolism. Journal of inherited metabolic disease. 2019;42(4):629-646.
39. Vrolijk MF et al. The vitamin B6 paradox: Supplementation with high concentrations of pyridoxine leads to decreased vitamin B6 function. Toxicol In Vitro. 2017;44:206-212.
40. Porter K et al. Causes, Consequences and Public Health Implications of Low B-Vitamin Status in Ageing. Nutrients. 2016;8(11).
41. Esse R, Barroso M, Tavares de Almeida I, Castro R. The Contribution of Homocysteine Metabolism Disruption to Endothelial Dysfunction: State-of-the-Art. International journal of molecular sciences. 2019;20(4).
42. Troesch B et al. Potential Links between Impaired One-Carbon Metabolism Due to Polymorphisms, Inadequate B-Vitamin Status, and the Development of Alzheimer's Disease. Nutrients. 2016;8(12).
43. Garcia-Minguillan CJ et al. Riboflavin status modifies the effects of methylenetetrahydrofolate reductase (MTHFR) and methionine synthase reductase (MTRR) polymorphisms on homocysteine. Genes Nutr. 2014;9(6):435.
44. Marashly ET et al. Riboflavin Has Neuroprotective Potential: Focus on Parkinson's Disease and Migraine. Frontiers in neurology. 2017;8:333.
45. Douaud G et al. Preventing Alzheimer's disease-related gray matter atrophy by B-vitamin treatment. Proc Natl Acad Sci U S A. 2013;110(23):9523-9528.
46. Mazza A et al. Nutraceutical approaches to homocysteine lowering in hypertensive subjects at low cardiovascular risk: a multicenter, randomized clinical trial. Journal of biological regulators and homeostatic agents. 2016;30(3):921-927.
47. Ueland PM. Choline and betaine in health and disease. Journal of inherited metabolic disease. 2011;34(1):3-15.
48. Craig SA. Betaine in human nutrition. The American Journal of Clinical Nutrition. 2004;80(3):539-549.
49. Zeisel S. Choline, Other Methyl-Donors and Epigenetics. Nutrients. 2017;9(5).
50. Obeid R. The metabolic burden of methyl donor deficiency with focus on the betaine homocysteine methyltransferase pathway. Nutrients. 2013;5(9):3481-3495.
51. Lee JE et al. Are dietary choline and betaine intakes determinants of total homocysteine concentration? Am J Clin Nutr. 2010;91(5):1303-1310.
52. Wallace TC et al. Assessment of Total Choline Intakes in the United States. J Am Coll Nutr. 2016;35(2):108-112.
53. Atkinson W et al. Dietary and supplementary betaine: effects on betaine and homocysteine concentrations in males. Nutr Metab Cardiovasc Dis. 2009;19(11):767-773.
54. Olthof MR et al. Effects of betaine intake on plasma homocysteine concentrations and consequences for health. Curr Drug Metab. 2005;6(1):15-22.
55. Steenge GR et al. Betaine supplementation lowers plasma homocysteine in healthy men and women. J Nutr. 2003;133(5):1291-1295.
56. Jakubowski H et al. Chemical biology of homocysteine thiolactone and related metabolites. Advances in clinical chemistry. 2011;55:81-103.
57. Cholewa JM et al. Effects of betaine on body composition, performance, and homocysteine thiolactone. Journal of the International Society of Sports Nutrition. 2013;10(1):39.
58. Guasch-Ferre M et al. Plasma Metabolites From Choline Pathway and Risk of Cardiovascular Disease in the PREDIMED (Prevention With Mediterranean Diet) Study. J Am Heart Assoc. 2017;6(11).
59. Meyer KA et al. Dietary Choline and Betaine and Risk of CVD: A Systematic Review and Meta-Analysis of Prospective Studies. Nutrients. 2017;9(7).
60. Rizzo G et al. The Link between Homocysteine and Omega-3 Polyunsaturated Fatty Acid: Critical Appraisal and Future Directions. Biomolecules. 2020;10(2).
61. Dawson SL et al. A combination of omega-3 fatty acids, folic acid and B-group vitamins is superior at lowering homocysteine than omega-3 alone: A meta-analysis. Nutr Res. 2016;36(6):499-508.
62. Jerneren F et al. Homocysteine Status Modifies the Treatment Effect of Omega-3 Fatty Acids on Cognition in a Randomized Clinical Trial in Mild to Moderate Alzheimer's Disease: The OmegAD Study. J Alzheimers Dis. 2019;69(1):189-197.
63. Hooper C et al. Cross-Sectional Associations of Total Plasma Homocysteine with Cortical beta-Amyloid Independently and as a Function of Omega 3 Polyunsaturated Fatty Acid Status in Older Adults at Risk of Dementia. The journal of nutrition, health & aging. 2017;21(10):1075-1080.
64. Jerneren F et al. Brain atrophy in cognitively impaired elderly: the importance of long-chain omega-3 fatty acids and B vitamin status in a randomized controlled trial. Am J Clin Nutr. 2015;102(1):215-221.
65. Oulhaj A et al. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J Alzheimers Dis. 2016;50(2):547-557.
66. Fu Y et al. Hyperhomocysteinaemia and vascular injury: advances in mechanisms and drug targets. Br J Pharmacol. 2018;175(8):1173-1189.
67. Bhatia P et al. Homocysteine excess: delineating the possible mechanism of neurotoxicity and depression. Fundamental & clinical pharmacology. 2015;29(6):522-528.
68. Papakostas GI et al. Folates and S-adenosylmethionine for major depressive disorder. Can J Psychiatry. 2012;57(7):406-413.
69. Karas Kuzelicki N. S-Adenosyl Methionine in the Therapy of Depression and Other Psychiatric Disorders. Drug Dev Res. 2016;77(7):346-356.
70. Anderson S et al. Anxiety and Methylenetetrahydrofolate Reductase Mutation Treated With S-Adenosyl Methionine and Methylated B Vitamins. Integrative medicine (Encinitas, Calif). 2016;15(2):48-52.
71. Mischoulon D et al. Bioavailability of S-adenosyl methionine and impact on response in a randomized, double-blind, placebo-controlled trial in major depressive disorder. The Journal of clinical psychiatry. 2012;73(6):843-848.
72. Aydin AF et al. N-Acetylcysteine supplementation decreased brain lipid and protein oxidations produced by experimental homocysteine thiolactone exposure: Relevance to neurodegeneration. Pathophysiology. 2018;25(2):125-129.
73. Kondakci G et al. The effect of N-acetylcysteine supplementation on serum homocysteine levels and hepatic and renal oxidative stress in homocysteine thiolactone-treated rats. Arch Physiol Biochem. 2017;123(2):128-133.
74. Yilmaz H et al. Effects of folic acid and N-acetylcysteine on plasma homocysteine levels and endothelial function in patients with coronary artery disease. Acta cardiologica. 2007;62(6):579-585.
75. Hildebrandt W et al. Oral N-acetylcysteine reduces plasma homocysteine concentrations regardless of lipid or smoking status. Am J Clin Nutr. 2015;102(5):1014-1024.
76. Shankle WR et al. CerefolinNAC Therapy of Hyperhomocysteinemia Delays Cortical and White Matter Atrophy in Alzheimer's Disease and Cerebrovascular Disease. J Alzheimers Dis. 2016;54(3):1073-1084.
77. Schaffer S et al. Effects and Mechanisms of Taurine as a Therapeutic Agent. Biomolecules & therapeutics. 2018;26(3):225-241.
78. Jakaria M et al. Taurine and its analogs in neurological disorders: Focus on therapeutic potential and molecular mechanisms. Redox biology. 2019;24:101223.
79. Zulli A et al. High dietary taurine reduces apoptosis and atherosclerosis in the left main coronary artery: association with reduced CCAAT/enhancer binding protein homologous protein and total plasma homocysteine but not lipidemia. Hypertension. 2009;53(6):1017-1022.
80. Nonaka H et al. Taurine prevents the decrease in expression and secretion of extracellular superoxide dismutase induced by homocysteine: amelioration of homocysteine-induced endoplasmic reticulum stress by taurine. Circulation. 2001;104(10):1165-1170.
81. Zhang Z et al. Taurine ameliorated homocysteine-induced H9C2 cardiomyocyte apoptosis by modulating endoplasmic reticulum stress. Apoptosis. 2017;22(5):647-661.
82. Ahn CS. Effect of taurine supplementation on plasma homocysteine levels of the middle-aged Korean women. Adv Exp Med Biol. 2009;643:415-422.
83. Van Hove JLK et al. Biomarkers of oxidative stress, inflammation, and vascular dysfunction in inherited cystathionine beta-synthase deficient homocystinuria and the impact of taurine treatment in a phase 1/2 human clinical trial. Journal of inherited metabolic disease. 2019;42(3):424-437.
84. Zhao L et al. Quantitative Association Between Serum/Dietary Magnesium and Cardiovascular Disease/Coronary Heart Disease Risk: A Dose-Response Meta-analysis of Prospective Cohort Studies. Journal of cardiovascular pharmacology. 2019;74(6):516-527.
85. Rosique-Esteban N et al. Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients. 2018;10(2).
86. Kirkland AE et al. The Role of Magnesium in Neurological Disorders. Nutrients. 2018;10(6).
87. Li W et al. Extracellular magnesium regulates effects of vitamin B6, B12 and folate on homocysteinemia-induced depletion of intracellular free magnesium ions in canine cerebral vascular smooth muscle cells: possible relationship to [Ca2+]i, atherogenesis and stroke. Neurosci Lett. 1999;274(2):83-86.
88. Guo H et al. Effects of folic acid and magnesium on the production of homocysteine-induced extracellular matrix metalloproteinase-2 in cultured rat vascular smooth muscle cells. Circulation journal. 2006;70(1):141-146.
89. Soni CV et al. Hyperhomocysteinemia Alters Sinoatrial and Atrioventricular Nodal Function: Role of Magnesium in Attenuating These Effects. Cell Biochem Biophys. 2016;74(1):59-65.
参考来源:
美国国立公众健康网
www.medlineplus.gov
美国国立心脏、肺、血液研究所
http://www.nhlbi.nih.gov
美国家庭医生学会
http://familydoctor.org
加拿大心血管协会
http://www.ccs.ca
加拿大家庭医生学会
http://www.cfpc.ca
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

